19. Chemical Thermodynamics
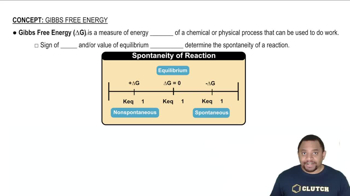
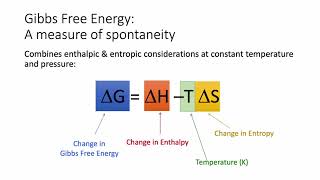
Gibbs Free Energy
Problem 62
Textbook Question
Textbook QuestionSulfur dioxide reacts with strontium oxide as follows: SO21g2 + SrO1g2¡SrSO31s2 (a) Without using thermochemical data, predict whether ΔG° for this reaction is more negative or less negative than ΔH°.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
634
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos