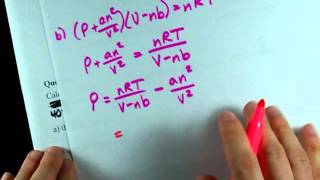7. Gases
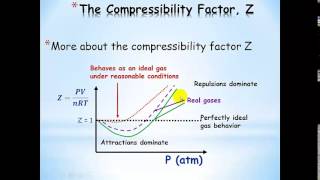
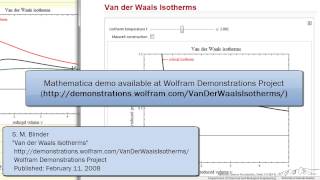
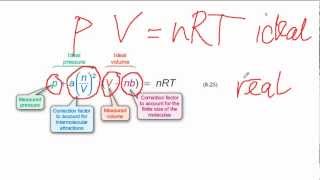
Van der Waals Equation
Problem 113b
Textbook Question
Textbook QuestionConsider the following gases, all at STP: Ne, SF6, N2, CH4. (d) Which one has the highest total molecular volume relative to the space occupied by the gas?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
586
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos