20. Electrochemistry
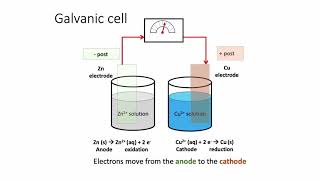
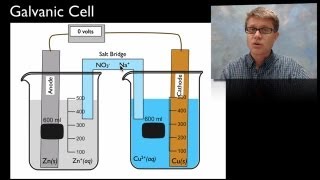
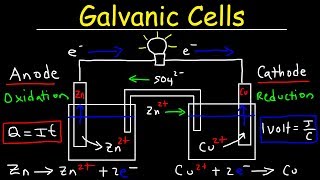
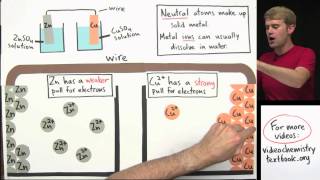
Galvanic Cell

Get help from an AI Tutor
Ask a question to get started.
Problem 29
Textbook Question
Textbook QuestionA voltaic cell similar to that shown in Figure 20.5 is constructed. One electrode half-cell consists of a silver strip placed in a solution of AgNO3, and the other has an iron strip placed in a solution of FeCl2. The overall cell reaction is Fe1s2 + 2 Ag+1aq2 ¡ Fe2+1aq2 + 2 Ag1s2 (f) In which directions do the cations and anions migrate through the solution?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
482
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos