19. Chemical Thermodynamics
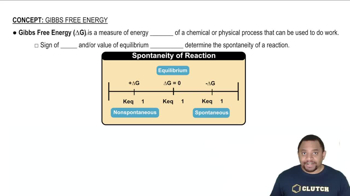
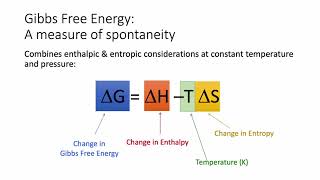
Gibbs Free Energy
Problem 37
Textbook Question
Textbook QuestionWithout doing any calculations, determine the signs of ΔSsys and ΔS surr for each chemical reaction. In addition, predict under what temperatures (all temperatures, low temperatures, or high temperatures), if any, the reaction is spontaneous. c. 2 N2( g) + O2( g) ¡ 2 N2O( g) ΔHrxn ° = +163.2 kJ
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
563
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos