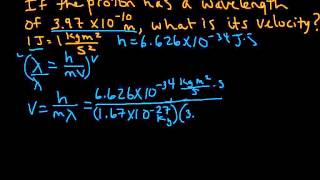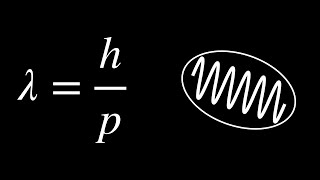9. Quantum Mechanics
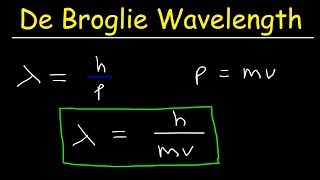
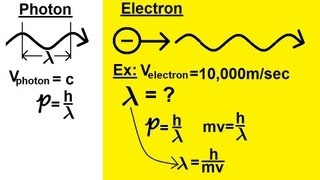
De Broglie Wavelength
Problem 133
Textbook Question
Textbook QuestionX rays with a wavelength of 1.54 * 10-10 m are produced when a copper metal target is bombarded with high-energy electrons that have been accelerated by a voltage difference of 30,000 V. The kinetic energy of the electrons equals the product of the voltage difference and the electronic charge in coulombs, where 1 volt-coulomb = 1 J. (a) What is the kinetic energy in joules and the de Broglie wavelength in meters of an electron that has been accel-erated by a voltage difference of 30,000 V?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
652
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos