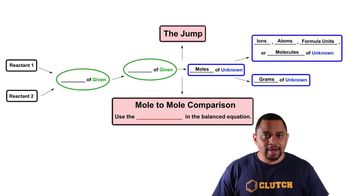
3. Chemical Reactions
Limiting Reagent
Problem 82a
Textbook Question
Textbook QuestionSolutions of sulfuric acid and lead(II) acetate react to form solid lead(II) sulfate and a solution of acetic acid. If 5.00 g of sulfuric acid and 5.00 g of lead(II) acetate are mixed, calculate the number of grams of lead(II) acetate present in the mixture after the reaction is complete.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
778
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos