15. Chemical Kinetics
Integrated Rate Law
Problem 128
Textbook Question
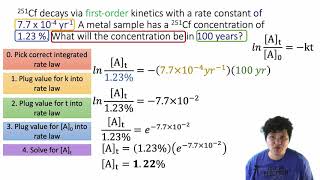
Textbook QuestionAssume that you are studying the first-order conversion of a reactant X to products in a reaction vessel with a constant volume of 1.000 L. At 1 p.m., you start the reaction at 25 °C with 1.000 mol of X. At 2 p.m., you find that 0.600 mol of X remains, and you immediately increase the temperature of the reaction mixture to 35 °C. At 3 p.m., you discover that 0.200 mol of X is still present. You want to finish the reaction by 4 p.m. but need to continue it until only 0.010 mol of X remains, so you decide to increase the temperature once again. What is the minimum temperature required to convert all but 0.010 mol of X to products by 4 p.m.?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
402
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos