2. Atoms & Elements
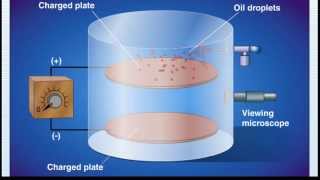
Millikan Oil Drop Experiment
Problem 87c
Textbook Question
Textbook QuestionSuppose a scientist repeats the Millikan oil-drop experiment but reports the charges on the drops using an unusual (and imaginary) unit called the warmomb (wa). The scientist obtains the following data for four of the drops: Droplet Calculated Charge (wa) A 3.84⨉10−8 B 4.80⨉10−8 C 2.88⨉10−8 D 8.64⨉10−8 (c) Based on your answer to part (b), how many electrons are there on each of the droplets?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
837
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos