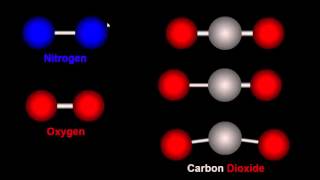
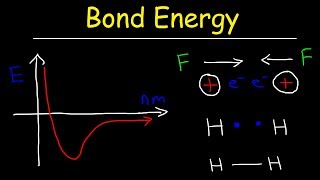
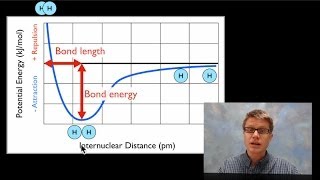
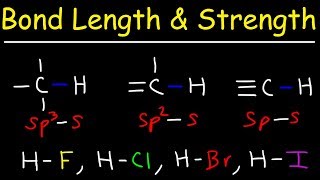11. Bonding & Molecular Structure
Bond Energy
Problem 28
Textbook Question
Textbook Question(b) Based on aver- age bond enthalpies, would you expect a photon capable of dissociating a C ¬ Cl bond to have sufficient energy to dis- sociate a C ¬ Br bond?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
316
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos