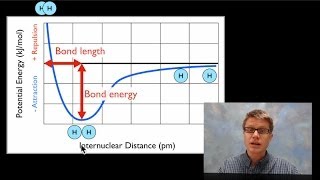
11. Bonding & Molecular Structure
Bond Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 111
Textbook Question
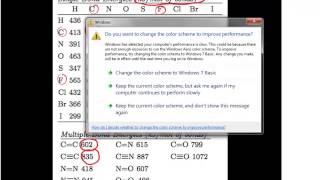
Textbook QuestionThe heat of atomization is the heat required to convert a molecule in the gas phase into its constituent atoms in the gas phase. The heat of atomization is used to calculate average bond energies. Without using any tabulated bond energies, calculate the average C¬Cl bond energy from the following data: the heat of atomization of CH4 is 1660 kJ>mol, and the heat of atomization of CH2Cl2 is 1495 kJ>mol.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1655
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos