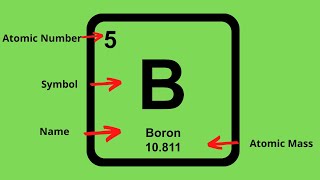
2. Atoms & Elements
Subatomic Particles
Problem 29c
Textbook Question
Textbook QuestionFill in the gaps in the following table, assuming each column represents a neutral atom. Symbol 159Tb Protons 29 37 Neutrons 34 53 Electrons 42 34 Mass no. 79 85 Complete the second row of the table excluding the row with isotope symbol.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
527
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos