7. Gases
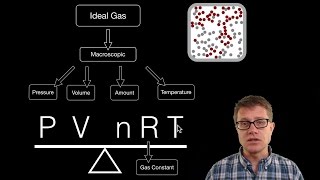
The Ideal Gas Law
Problem 40a
Textbook Question
Textbook QuestionAn aerosol spray can with a volume of 125 mL contains 1.30 g of propane gas (C3H8) as a propellant. (c) The can's label says that exposure to temperatures above 50 °C may cause the can to burst. What is the pressure in the can at this temperature?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
1029
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos