13. Liquids, Solids & Intermolecular Forces
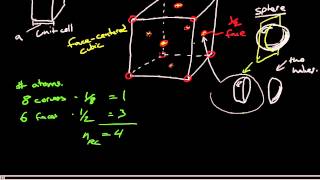
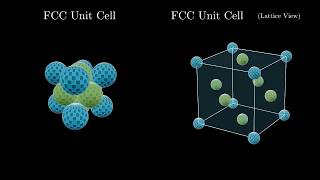
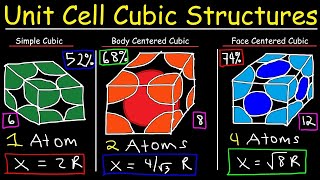
Face Centered Cubic Unit Cell

Get help from an AI Tutor
Ask a question to get started.
Problem 42
Textbook Question
Textbook QuestionTitanium metal has a density of 4.506 g>cm3 and an atomic radius of 144.8 pm. In what cubic unit cell does titanium crystallize?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
859
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos