8. Thermochemistry
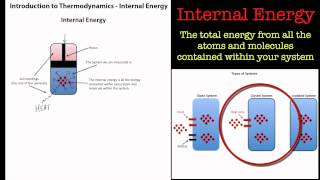
Internal Energy
Problem 56b
Textbook Question
Textbook QuestionThe change in internal energy for the combustion of 1.0 mol of octane at a pressure of 1.0 atm is 5084.3 kJ. If the change in enthalpy is 5074.1 kJ, how much work is done during the combustion?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
2132
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos