Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (e) aluminum metal is added to a solution of cobalt(II) sulfate.

Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (d) chromium metal is immersed in an aqueous solution of cobalt(II) chloride
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Activity Series
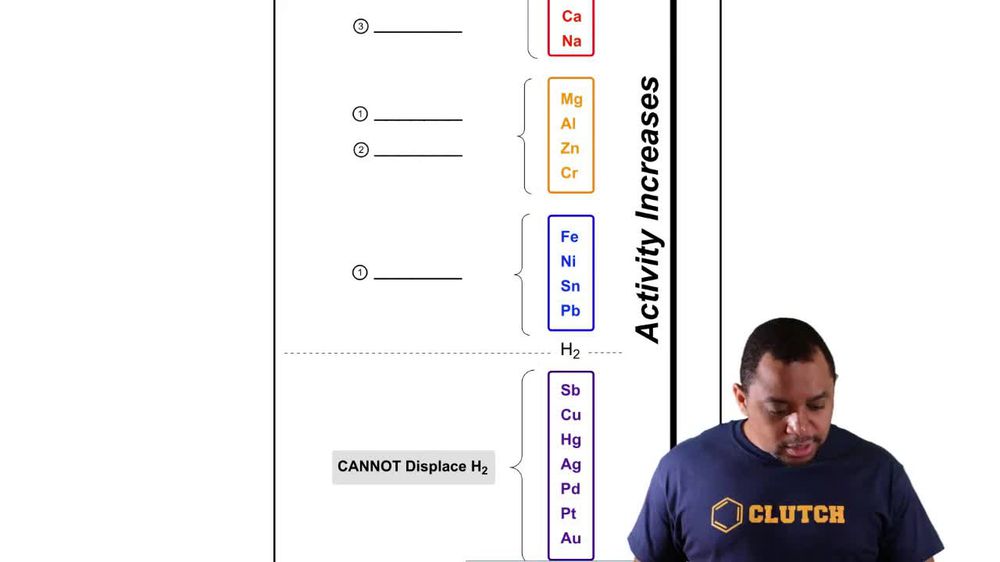
Redox Reactions

Balanced Chemical Equations

Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (a) Nickel metal is added to a solution of copper(II) nitrate
Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (b) a solution of zinc nitrate is added to a solution of magnesium sulfate (c) hydrochloric acid is added to gold metal
Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (e) hydrogen gas is bubbled through a solution of silver nitrate.
The metal cadmium tends to form Cd2+ ions. The following observations are made: (i) When a strip of zinc metal is placed in CdCl2(aq), cadmium metal is deposited on the strip. (ii) When a strip of cadmium metal is placed in Ni(NO3)(aq), nickel metal is deposited on the strip. (a) Write net ionic equations to explain each of the preceding observations.
The metal cadmium tends to form Cd2+ ions. The following observations are made: (i) When a strip of zinc metal is placed in CdCl2(aq), cadmium metal is deposited on the strip. (ii) When a strip of cadmium metal is placed in Ni(NO3)(aq), nickel metal is deposited on the strip. (b) Which elements more closely define the position of cadmium in the activity series?
