2. Atoms & Elements
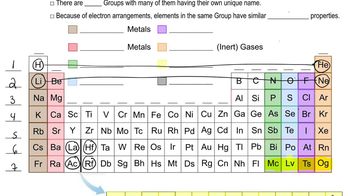
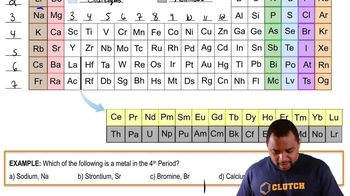
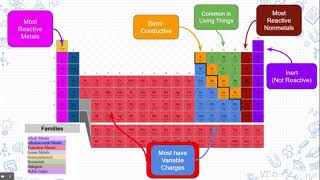
Periodic Table: Group Names
Problem 64
Textbook Question
Textbook QuestionList several general properties of the following groups: (a) Alkali metals (b) Noble gases (c) Halogens
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
794
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos