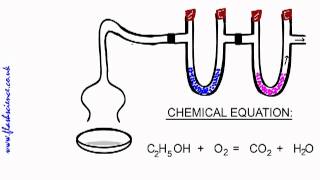
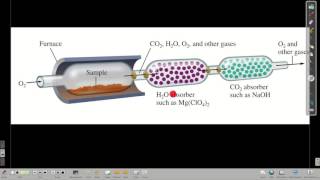
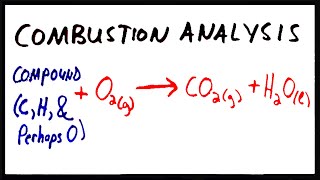
3. Chemical Reactions
Combustion Analysis
Problem 107
Textbook Question
Textbook Question(a) Combustion analysis of 150.0 mg of 1,2,3,benzenetriol, a compound composed of carbon, hydrogen, and oxy-gen, gives 64.3 mg of H2O and 314.2 mg of CO2. What is the empirical formula of 1,2,3,benzenetriol?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
425
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos