2. Atoms & Elements
Periodic Table: Element Symbols
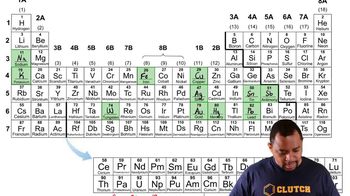
Problem 54b
Textbook Question
Textbook QuestionExamine Figure 2.2, A portion of Mendeleev's periodic table. (a) Which characteristic was used to organize the elements in the table?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
43sPlay a video:
351
views
Was this helpful?
Related Videos
Related Practice