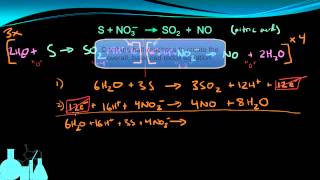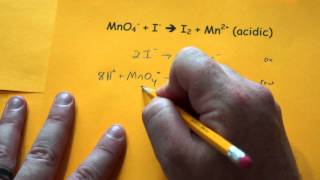6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions

Get help from an AI Tutor
Ask a question to get started.
Problem 106
Textbook Question
Textbook QuestionConsider the unbalanced redox reaction: Cr2O72-(aq) + Cu(s) ¡ Cr3+(aq) + Cu2+(aq) Balance the equation and determine the volume of a 0.850 M K2Cr2O7 solution required to completely react with 5.25 g of Cu.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
1807
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos