13. Liquids, Solids & Intermolecular Forces
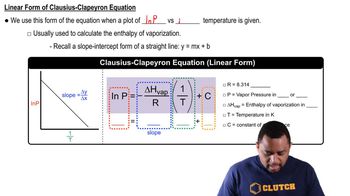
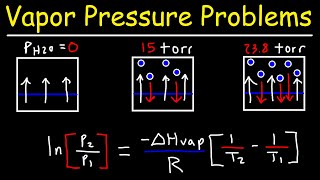
Clausius-Clapeyron Equation
Problem 65
Textbook Question
Textbook QuestionCarbon disulfide has a vapor pressure of 363 torr at 25 °C and a normal boiling point of 46.3 °C. Find ΔHvap for carbon disulfide.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
2422
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos