8. Thermochemistry
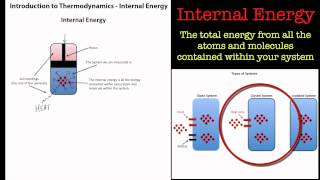
Internal Energy
Problem 56a
Textbook Question
Textbook QuestionWhen a sample of a hydrocarbon fuel is ignited and burned in oxygen, the internal energy decreases by 7.20 kJ. If 5670 J of heat were transferred to the surroundings, what is the sign and magnitude of work? If the reaction took place in an environ- ment with a pressure of 1 atm, what was the volume change?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
11mPlay a video:
906
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos