1. Intro to General Chemistry
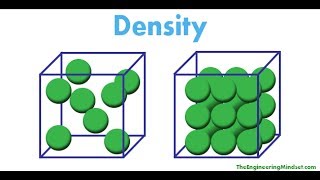
Density
Problem 70a
Textbook Question
Textbook QuestionGaseous hydrogen has a density of 0.0899 g/L at 0 °C, and gaseous chlorine has a density of 3.214 g/L at the same tem-perature. How many liters of each would you need if you wanted 1.0078 g of hydrogen and 35.45 g of chlorine?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1135
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos