19. Chemical Thermodynamics
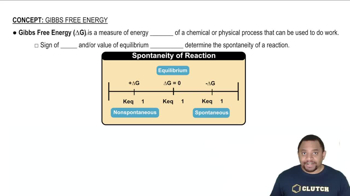
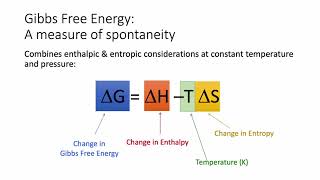
Gibbs Free Energy
Problem 63a
Textbook Question
Textbook QuestionClassify each of the following reactions as one of the four possible types summarized in Table 19.3: (i) spontanous at all temperatures; (ii) not spontaneous at any temperature; (iii) spontaneous at low T but not spontaneous at high T; (iv) spontaneous at high T but not spontaneous at low T. (c) N2F41g2¡2 NF21g2 ΔH° = 85 kJ; ΔS° = 198 J>K
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
823
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos