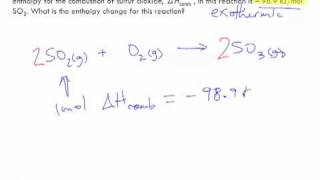8. Thermochemistry
Thermochemical Equations

Get help from an AI Tutor
Ask a question to get started.
Problem 69
Textbook Question
Textbook QuestionHow much energy in kilojoules is required to convert 100 mL of water at its boiling point from liquid to vapor, and how does this compare with the result calculated in Problem 9.68 for diethyl ehter? [ΔHvap(H2O) = + 40.7 kJ/mol]
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
571
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos