Textbook Question
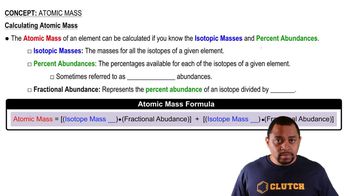
Naturally occurring silver consists of two isotopes: 107^Ag (51.84%) with an isotopic mass of 106.9051 and 109^Ag (48.16%) with an isotopic mass of 108.9048. What is the atomic weight of silver? Check your answer in a periodic table.
3104
views