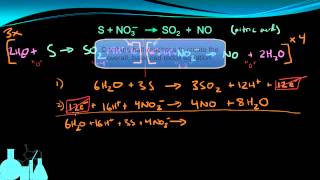6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions
Problem 51a
Textbook Question
Textbook QuestionGiven the following reduction half-reactions: Fe3+1aq2 + e- ¡ Fe2+1aq2 E°red = +0.77 V S2O62 - 1aq2 + 4 H+1aq2 + 2 e- ¡ 2 H2SO31aq2 E°red = +0.60 V N2O1g2 + 2 H+1aq2 + 2 e- ¡ N21g2 + H2O1l2 Ered ° = -1.77 V VO2+1aq2 + 2 H+1aq2 + e- ¡ VO2+ + H2O1l2 E°red = +1.00 V (a) Write balanced chemical equations for the oxidation of Fe2+1aq2 by S2O62-1aq2, by N2O1aq2, and by VO2+1aq2.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
11mPlay a video:
716
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos