2. Atoms & Elements
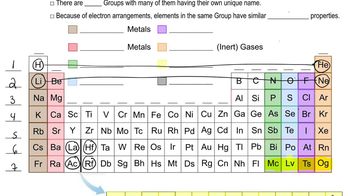
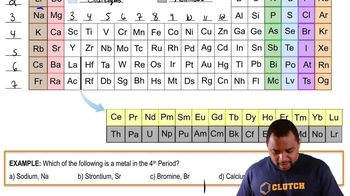
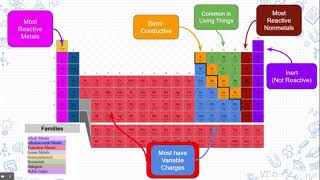
Periodic Table: Group Names
Problem 66
Textbook Question
Textbook QuestionWithout looking at a periodic table, list as many halogens as you can. (There are four common ones.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
291
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos