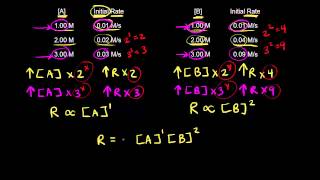15. Chemical Kinetics
Rate Law
Problem 93
Textbook Question
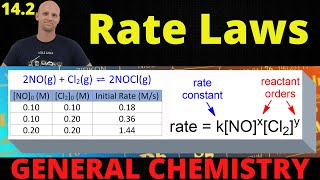
Textbook QuestionConsider the following reaction between mercury(II) chloride and oxalate ion: 2 HgCl21aq2 + C2O4 2 - 1aq2¡2 Cl - 1aq2 + 2 CO21g2 + Hg2Cl21s2 The initial rate of this reaction was determined for several concentrations of HgCl2 and C2O4 2 -, and the following rate data were obtained for the rate of disappearance of C2O4 2 - : Experiment 3HgCl2 4 1M 2 3C2o4 24 1M 2 Rate 1M,s2 1 0.164 0.15 3.2 * 10-5 2 0.164 0.45 2.9 * 10-4 3 0.082 0.45 1.4 * 10-4 4 0.246 0.15 4.8 * 10-5 (c) What is the reaction rate when the initial concentration of HgCl2 is 0.100 M and that of C2O4 2- is 0.25 M if the temperature is the same as that used to obtain the data shown?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1226
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos