11. Bonding & Molecular Structure
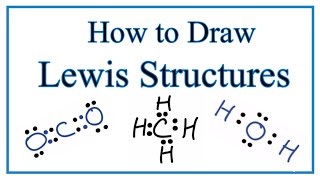
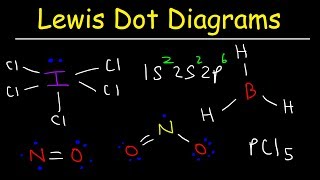
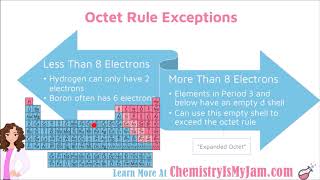
Lewis Dot Structures: Exceptions
Problem 64
Textbook Question
Textbook QuestionDraw the Lewis structures for each of the following molecules or ions. Identify instances where the octet rule is not obeyed; state which atom in each compound does not follow the octet rule; and state how many electrons surround these atoms: (a) PF6-, (b) BeCl2, (c) NH3, (d) XeF2O (the Xe is the central atom), (e) SO42 - .
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1239
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos