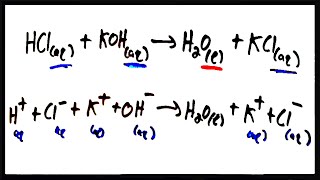6. Chemical Quantities & Aqueous Reactions
Molecular Equations
Problem 91b
Textbook Question
Textbook QuestionUranium hexafluoride, UF6, is processed to produce fuel for nuclear reactors and nuclear weapons. UF6 can be produced in a two-step reaction. Solid uranium (IV) oxide, UO2, is first made to react with hydrofluoric acid (HF) solution to form solid UF4 with water as a by-product. UF4 further reacts with fluorine gas to form UF6. (b) Which step is an acid-base reaction?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
915
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos