Consider the phase diagram shown here. Identify the states present at points a through g.
Ch.11 - Liquids, Solids & Intermolecular Forces

Chapter 11, Problem 76
Argon has a normal boiling point of 87.2 K and a melting point (at 1 atm) of 84.1 K. Its critical temperature is 150.8 K and its critical pressure is 48.3 atm. It has a triple point at 83.7 K and 0.68 atm. Sketch the phase diagram for argon. Which has the greater density, solid argon or liquid argon?
 Verified step by step guidance
Verified step by step guidance1
Identify the key points for the phase diagram: the triple point (83.7 K, 0.68 atm), the normal boiling point (87.2 K, 1 atm), the melting point (84.1 K, 1 atm), and the critical point (150.8 K, 48.3 atm).
Draw the axes for the phase diagram with pressure on the y-axis and temperature on the x-axis.
Plot the triple point, normal boiling point, melting point, and critical point on the diagram.
Draw the phase boundaries: the line from the triple point to the melting point represents the solid-liquid equilibrium, the line from the triple point to the normal boiling point represents the solid-gas equilibrium, and the line from the normal boiling point to the critical point represents the liquid-gas equilibrium.
To determine which phase has greater density, consider that typically, the solid phase is denser than the liquid phase, but for some substances like water, the liquid phase can be denser. Compare the slopes of the solid-liquid equilibrium line to infer density differences.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
6mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Phase Diagram
A phase diagram is a graphical representation that shows the states of a substance (solid, liquid, gas) at various temperatures and pressures. It includes key points such as the melting point, boiling point, critical point, and triple point, which indicate the conditions under which different phases coexist. Understanding how to read and interpret a phase diagram is essential for analyzing the behavior of substances under varying conditions.
Recommended video:
Guided course
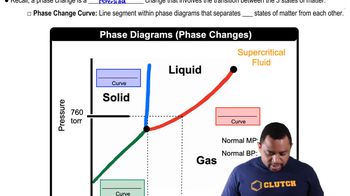
Phase Changes in Diagrams
Density of Phases
Density is defined as mass per unit volume and varies between different phases of a substance. Generally, solids are denser than liquids due to their closely packed molecular structure. In the case of argon, comparing the densities of solid and liquid phases involves understanding how temperature and pressure affect molecular arrangement and spacing, which is crucial for determining which phase has greater density.
Recommended video:
Guided course
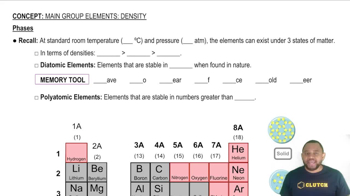
Phases
Triple Point
The triple point of a substance is the unique set of conditions at which all three phases (solid, liquid, and gas) coexist in equilibrium. For argon, the triple point occurs at 83.7 K and 0.68 atm, indicating the specific temperature and pressure where solid, liquid, and gaseous argon can exist simultaneously. This concept is vital for understanding phase transitions and the stability of different states of matter.
Recommended video:
Guided course

Boiling Point Elevation
Related Practice
Textbook Question
1221
views
Textbook Question
Nitrogen has a normal boiling point of 77.3 K and a melting point (at 1 atm) of 63.1 K. Its critical temperature is 126.2 K and its critical pressure is 2.55×104 torr. It has a triple point at 63.1 K and 94.0 torr. Sketch the phase diagram for nitrogen. Does nitrogen have a stable liquid state at 1 atm?
2550
views
Textbook Question
The phase diagram for sulfur is shown here. The rhombic and monoclinic states are two solid states with different structures. a. Below what pressure does solid sulfur sublime?
1585
views
Textbook Question
The phase diagram for sulfur is shown here. The rhombic and monoclinic states are two solid states with different structures. b. Which of the two solid states of sulfur is more dense?
525
views
