A compound with the formula XOCl2 reacts with water, yielding HCl and another acid H2XO3, which has two acidic hydrogens that react with NaOH. When 0.350 g of XOCl2 was added to 50.0 mL of water and the resultant solution was titrated, 96.1 mL of 0.1225 M NaOH was required to react with all the acid. (a) Write a balanced equation for the reaction of XOCl2 with H2O.

Some metals occur naturally in their elemental state while others occur as compounds in ores. Gold, for instance, is found as the free metal; mercury is obtained by heating mercury(II) sulfide ore in oxygen; and zinc is obtained by heating zinc(II) oxide ore with coke (carbon). Judging from their positions in the activity series, which of the metals sil-ver, platinum, and chromium would probably be obtained by (a) finding it in its elemental state?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Activity Series of Metals
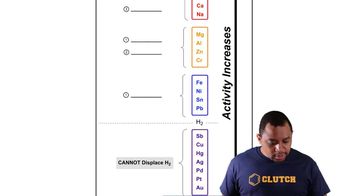
Elemental State of Metals

Reduction Processes in Metallurgy

A compound with the formula XOCl2 reacts with water, yielding HCl and another acid H2XO3, which has two acidic hydrogens that react with NaOH. When 0.350 g of XOCl2 was added to 50.0 mL of water and the resultant solution was titrated, 96.1 mL of 0.1225 M NaOH was required to react with all the acid. (b) What are the atomic mass and identity of element X?
Some metals occur naturally in their elemental state while others occur as compounds in ores. Gold, for instance, is found as the free metal; mercury is obtained by heating mercury(II) sulfide ore in oxygen; and zinc is obtained by heating zinc(II) oxide ore with coke (carbon). Judging from their positions in the activity series, which of the metals sil-ver, platinum, and chromium would probably be obtained by (c) heating its oxide with coke?
(b) If Ksp = 1.1 * 10-12 for Ag2CrO4, what are the molar concentrations of Ag+ and CrO4 2-in solution?
