7. Gases
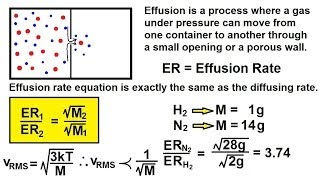
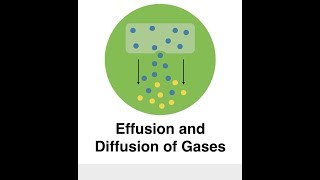
Effusion
Problem 85b
Textbook Question
Textbook QuestionWe separate U-235 from U-238 by fluorinating a sample of uranium to form UF6 (which is a gas) and then taking advantage of the different rates of effusion and diffusion for compounds containing the two isotopes. Calculate the ratio of effusion rates for 238UF6 and 235UF6. The atomic mass of U-235 is 235.054 amu and that of U-238 is 238.051 amu.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
2161
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos