2. Atoms & Elements
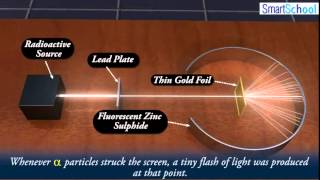
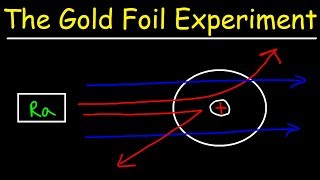
Rutherford Gold Foil Experiment
Problem 108
Textbook Question
Textbook QuestionThe molecular weight of ethylene glycol is 62.0689 when calculated using the atomic weights found in a standard periodic table, yet the molecular weight determined experimentally by high-resolution mass spectrometry is 62.0368. Explain the discrepancy.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1644
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos