8. Thermochemistry
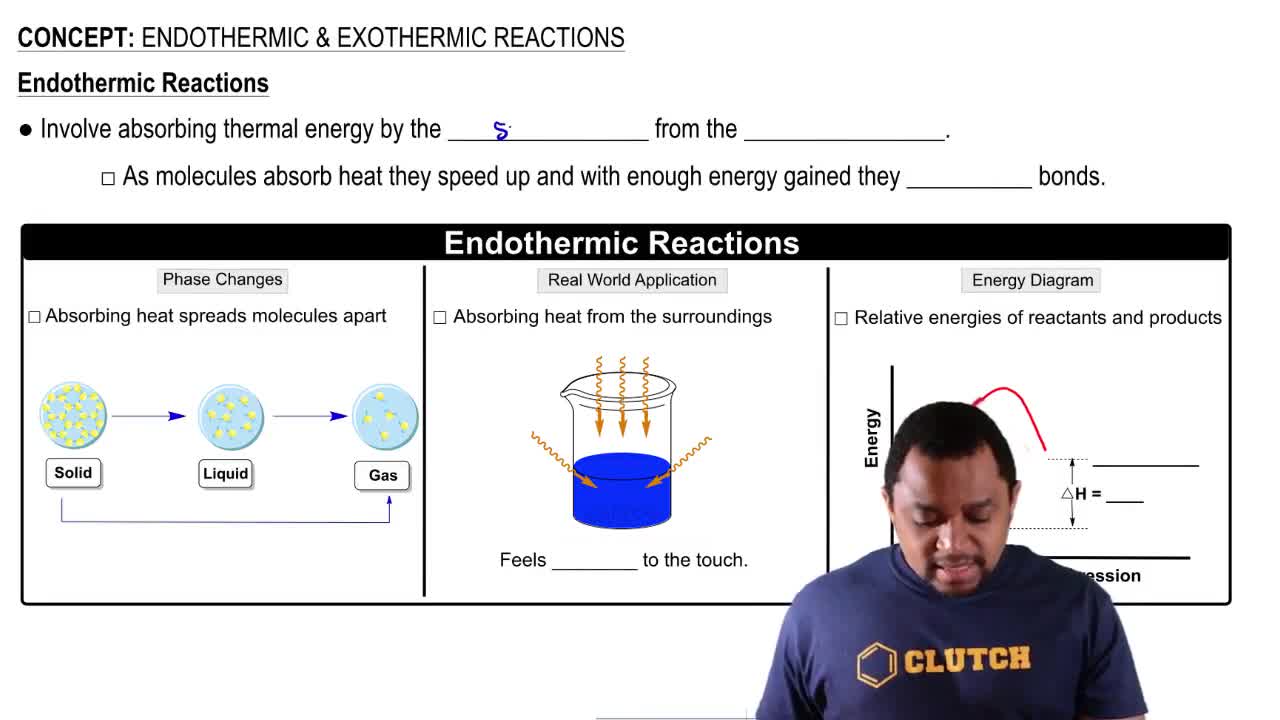
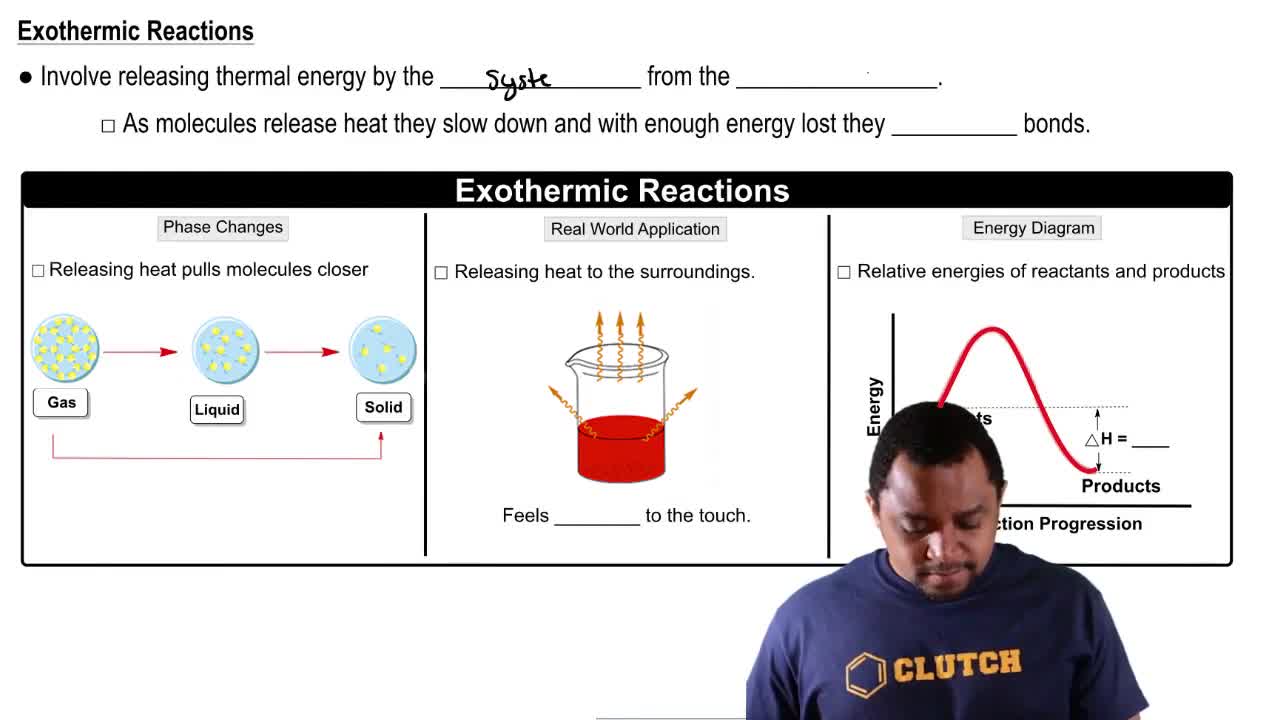
Endothermic & Exothermic Reactions
Problem 46
Textbook Question
Textbook QuestionAt one time, a common means of forming small quantities of oxygen gas in the laboratory was to heat KClO3: 2 KClO31s2¡2 KCl1s2 + 3 O21g2 H = -89.4 kJ (c) The decomposition of KClO3 proceeds spontaneously when it is heated. Do you think that the reverse reaction, the formation of KClO3 from KCl and O2, is likely to be feasible under ordinary conditions? Explain your answer.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
477
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos