8. Thermochemistry
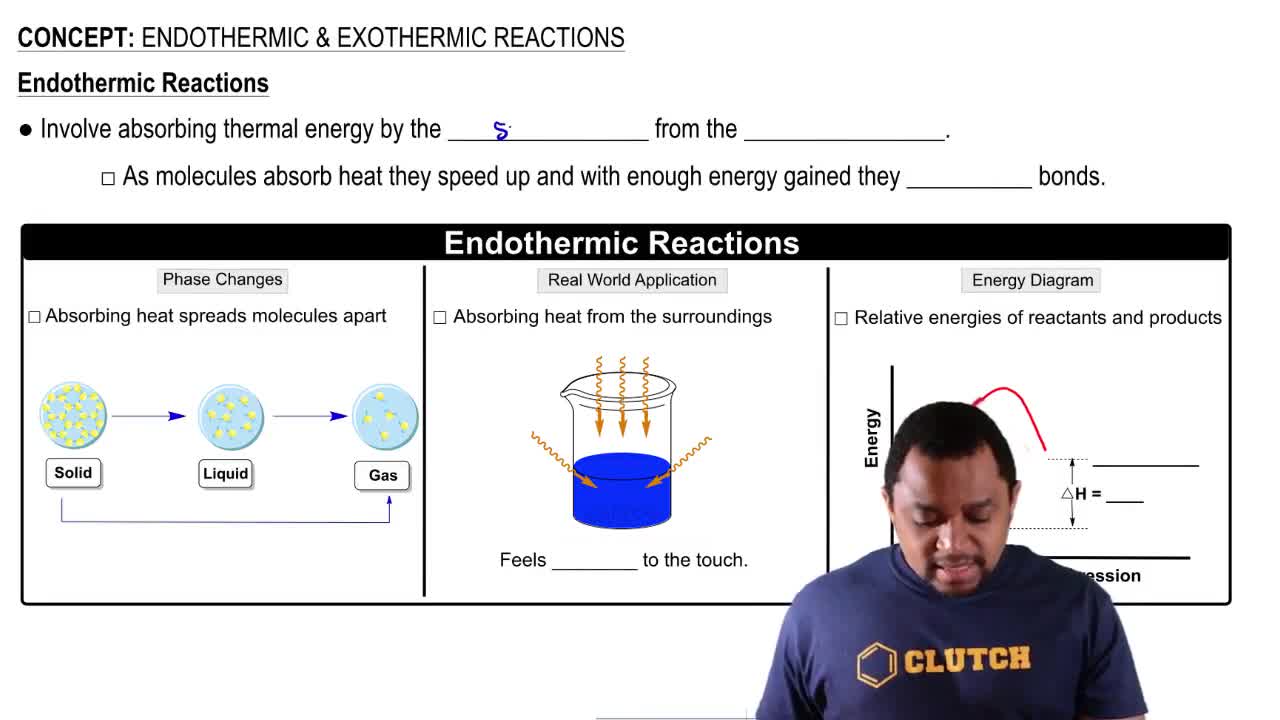
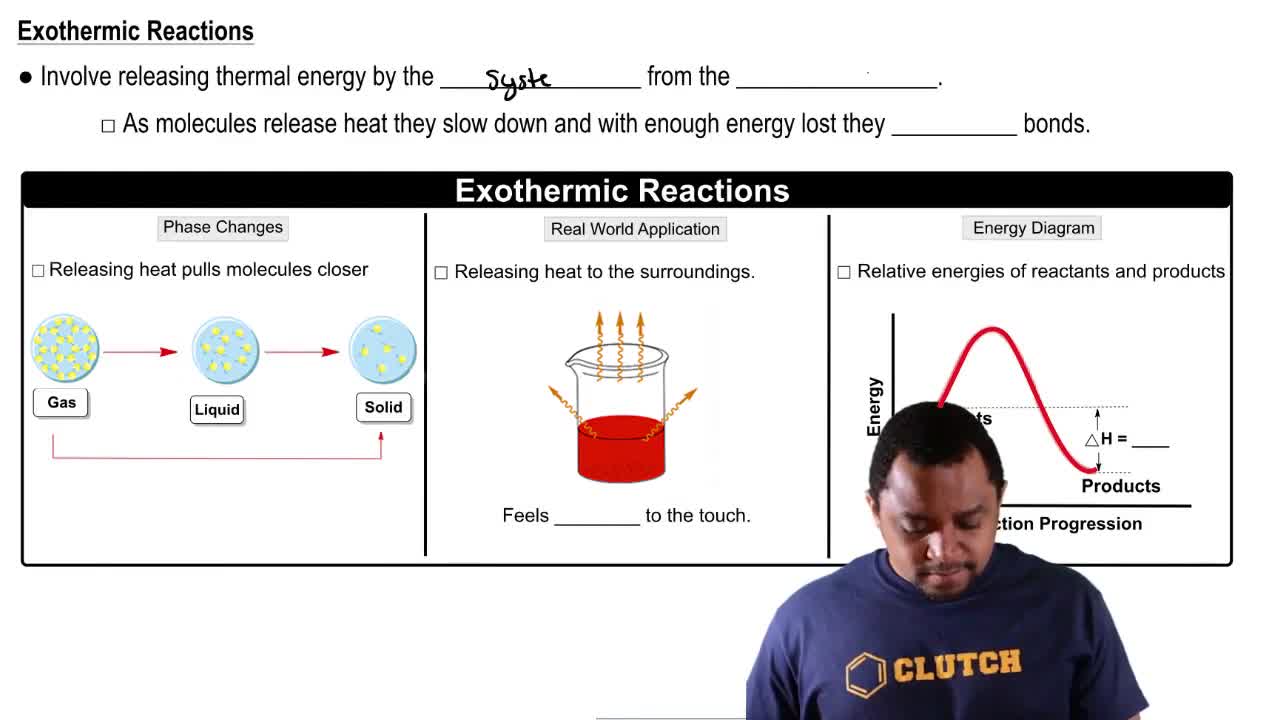
Endothermic & Exothermic Reactions
Problem 8
Textbook Question
Textbook QuestionImagine a container placed in a tub of water, as depicted in the accompanying diagram. (a) If the contents of the container are the system and heat is able to flow through the container walls, what qualitative changes will occur in the temperatures of the system and in its surroundings? From the system's perspective, is the process exothermic or endothermic?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
287
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos