2. Atoms & Elements
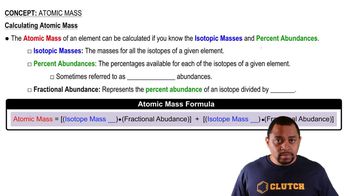
Atomic Mass

Get help from an AI Tutor
Ask a question to get started.
Problem 100
Textbook Question
Textbook QuestionNaturally occurring iodine has an atomic mass of 126.9045 amu. A 12.3849 g sample of iodine is accidentally contaminated with an additional 1.00070 g of 129I, a synthetic radioisotope of iodine used in the treatment of certain diseases of the thyroid gland. The mass of 129I is 128.9050 amu. Find the apparent 'atomic mass' of the contaminated iodine.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1426
views
2
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos