3. Chemical Reactions
Empirical Formula
Problem 97
Textbook Question
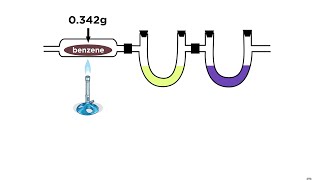
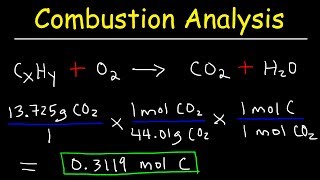
Textbook QuestionCombustion analysis of a hydrocarbon produces 33.01 g CO2 and 13.51 g H2O. Calculate the empirical formula of the hydrocarbon.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
4689
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos