6. Chemical Quantities & Aqueous Reactions
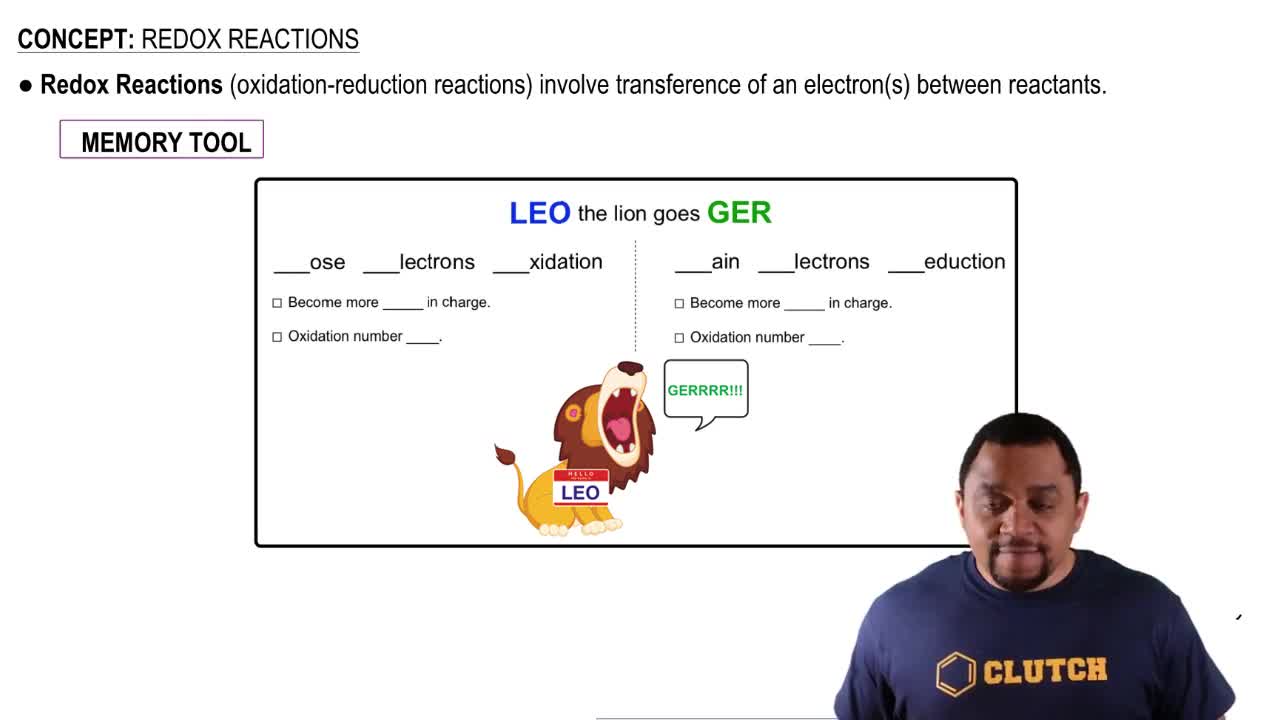
Redox Reactions

Get help from an AI Tutor
Ask a question to get started.
Problem 100c
Textbook Question
Textbook QuestionSilver can be electroplated at the cathode of an electrolysis cell by the half-reaction: Ag+(aq) + e- ¡ Ag(s) What mass of silver would plate onto the cathode if a current of 6.8 A flowed through the cell for 72 min?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1739
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos