Balance the following equations:
(a) HClO4(aq) + P4O10(s) → HPO3(aq) + Cl2O7(l)
(b) Au2S3(s + H2(g) → Au(s) + H2S(g)

 Verified step by step guidance
Verified step by step guidance
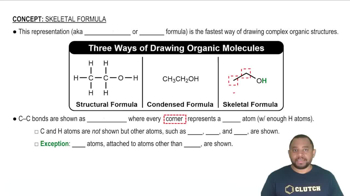


Balance the following equations:
(a) HClO4(aq) + P4O10(s) → HPO3(aq) + Cl2O7(l)
(b) Au2S3(s + H2(g) → Au(s) + H2S(g)
Write 'true' or 'false' for each statement. (a) We balance chemical equations as we do because energy must be conserved.
Write 'true' or 'false' for each statement. (b) If the reaction 2 O3(g) → 3 O2(g) goes to completion and all O3 is converted to O2, then the mass of O3 at the beginning of the reaction must be the same as the mass of O2 at the end of the reaction.
Balance the following equations: (c) Al(OH)3(s) + H2SO4(l) → Al2(SO4)3(s) + H2O(l)
Nitrogen monoxide and oxygen react to form nitrogen dioxide. Consider the mixture of NO and O2 shown in the accompanying diagram. The blue spheres represent N, and the red ones represent O. (c) If the actual yield of the reaction was 75% instead of 100%, how many molecules of each kind would be present after the reaction was over?