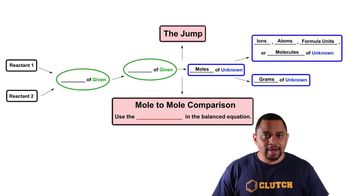
3. Chemical Reactions
Limiting Reagent
Problem 85a
Textbook Question
Textbook QuestionSodium borohydride, NaBH4, a substance used in the synthesis of many pharmaceutical agents, can be prepared by reaction of NaH with B2H6 according to the equation 2 NaH + B2H6 ---> 2 NaBH4 (b) Which reactant is limiting, and how many grams of the excess reactant will be left over?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
718
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos