8. Thermochemistry
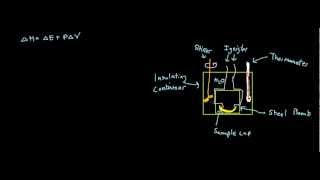
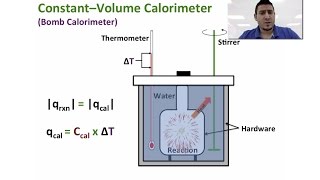
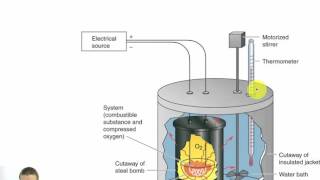
Constant-Volume Calorimetry
Problem 93
Textbook Question
Textbook QuestionThe heat of combustion of fructose, C6H12O6, is -2812 kJ>mol. If a fresh golden delicious apple weighing 120 g contains 16.0 g of fructose, what caloric content does the fructose contribute to the apple?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1364
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos