6. Chemical Quantities & Aqueous Reactions
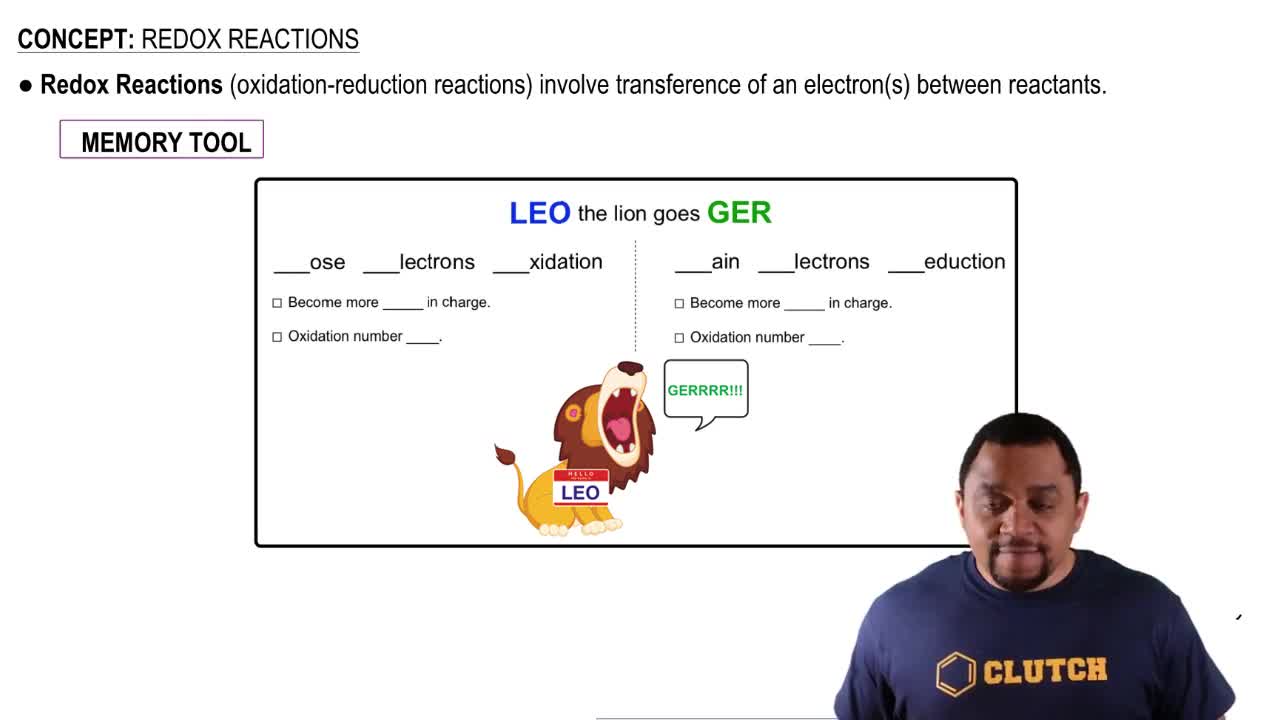
Redox Reactions
Problem 100
Textbook Question
Textbook QuestionGold exists in two common positive oxidation states, +1 and +3. The standard reduction potentials for these oxidation states are Au+1aq2 + e- ¡ Au1s2 Ered ° = +1.69 V Au3+1aq2 + 3 e- ¡ Au1s2 Ered ° = +1.50 V (c) Miners obtain gold by soaking gold-containing ores in an aqueous solution of sodium cyanide. A very soluble complex ion of gold forms in the aqueous solution because of the redox reaction 4 Au1s2 + 8 NaCN1aq2 + 2 H2O1l2 + O21g2 ¡ 4 Na3Au1CN2241aq2 + 4 NaOH1aq2 What is being oxidized, and what is being reduced in this reaction?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
1654
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos