Textbook Question
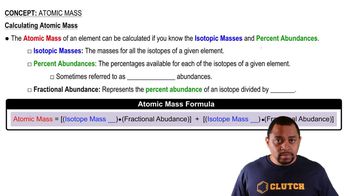
Magnesium has three naturally occurring isotopes: 24Mg (23.985) with 78.99% abundance, 25Mg (24.986) with 10.00% abundance, and a third with 11.01% abundance. Look up the atomic weight of magnesium, and then calculate the mass of the third isotope.
6869
views
1
rank
1
comments