14. Solutions
Solutions: Solubility and Intermolecular Forces

Get help from an AI Tutor
Ask a question to get started.
Problem 2b
Textbook Question
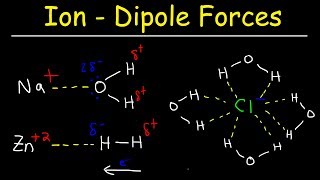
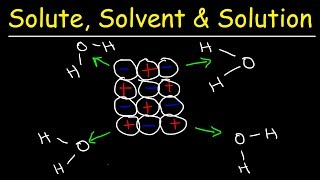
Textbook QuestionThis figure shows the interaction of a cation with surrounding water molecules. (b) Which of the following explanations accounts for the fact that the ion–solvent interaction is greater for Li+ than for K+? a. Li+ is of lower mass than K+. b. The ionization energy of Li is higher than that for K. c. Li+ has a smaller ionic radius than K+. d. Li has a lower density than K. e. Li reacts with water more slowly than K. [Section 13.1]
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
273
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos