6. Chemical Quantities & Aqueous Reactions
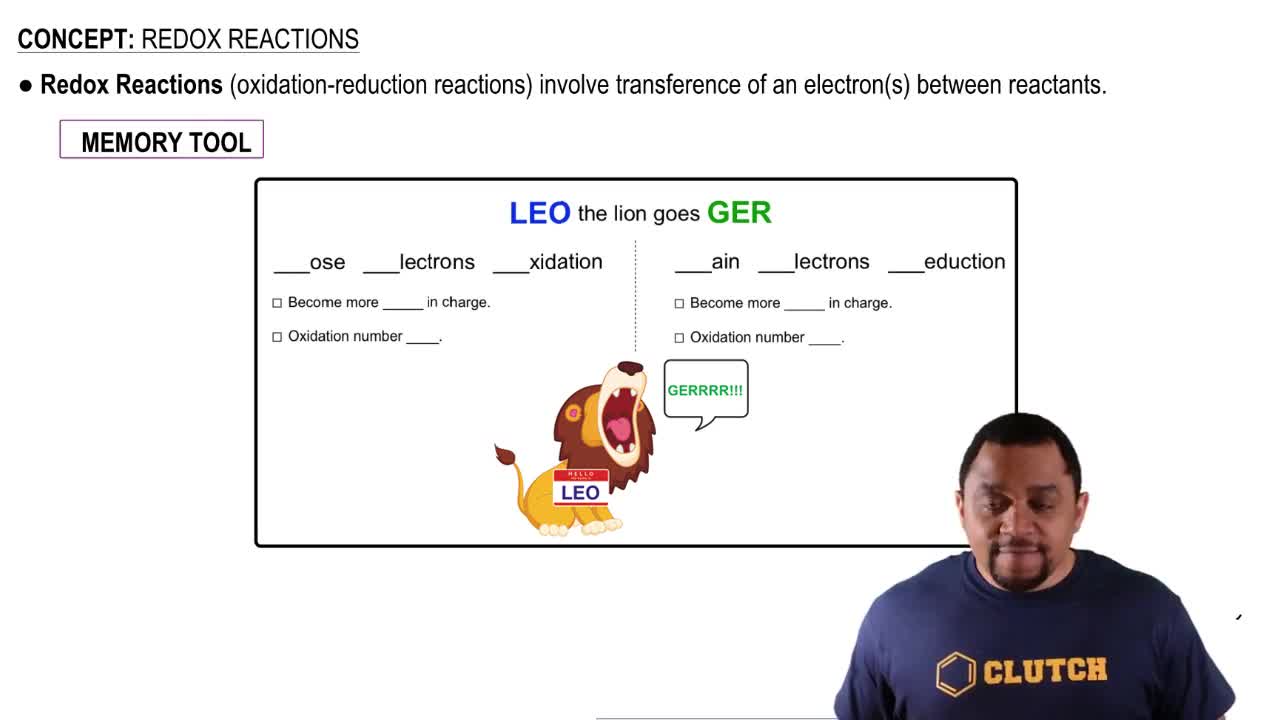
Redox Reactions
Problem 98
Textbook Question
Textbook QuestionDetermine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq)¡Al3 + (aq) + 3 Ag(s) b. SO3( g) + H2O(l )¡H2SO4(aq) c. Ba(s) + Cl2( g)¡BaCl2(s) d. Mg(s) + Br2(l )¡MgBr2(s)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
417
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos