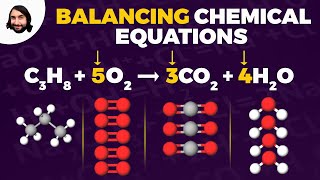
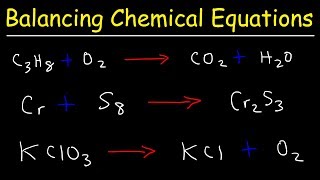
3. Chemical Reactions
Balancing Chemical Equations
Problem 16d
Textbook Question
Textbook QuestionWrite balanced chemical equations to correspond to each of the following descriptions: (d) When solid mercury(II) nitrate is heated, it decomposes to form solid mercury(II) oxide, gaseous nitrogen dioxide, and oxygen.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
796
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos