1. Intro to General Chemistry
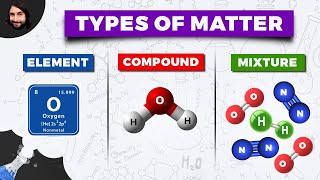
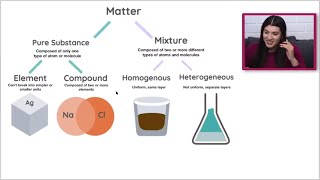
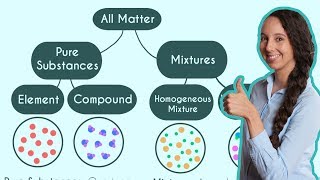
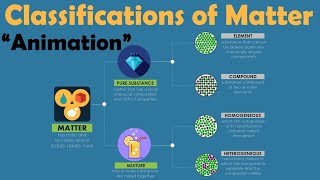
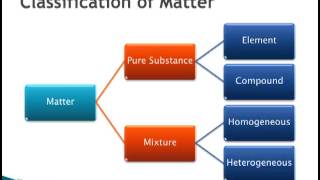
Classification of Matter
Problem 65a
Textbook Question
Textbook QuestionClassify each of the following as a pure substance, a solution, or a heterogeneous mixture: (b) a 999 gold bar
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1005
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 17 videos