13. Liquids, Solids & Intermolecular Forces
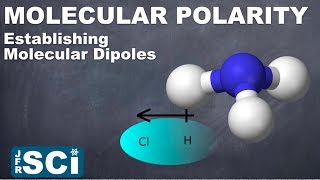
Molecular Polarity
Problem 85
Textbook Question
Textbook QuestionFluorine is more electronegative than chlorine, yet fluoromethane 1CH3F; m = 1.86 D2 has a smaller dipole moment than chloromethane 1CH3Cl; m = 1.90 D2. Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
836
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos