3. Chemical Reactions
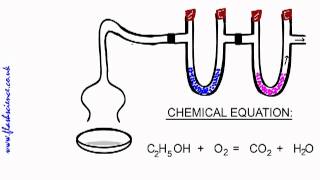
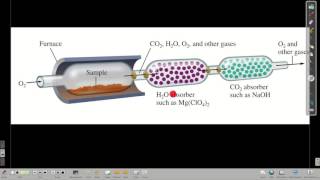
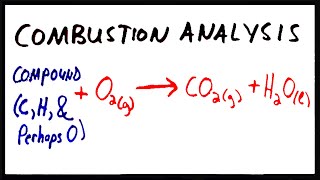
Combustion Analysis
Problem 100
Textbook Question
Textbook QuestionTartaric acid is the white, powdery substance that coats tart candies such as Sour Patch Kids. Combustion analysis of a 12.01-g sample of tartaric acid—which contains only carbon, hydrogen, and oxygen—produces 14.08 g CO2 and 4.32 g H2O. Determine the empirical formula of tartaric acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
2203
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos